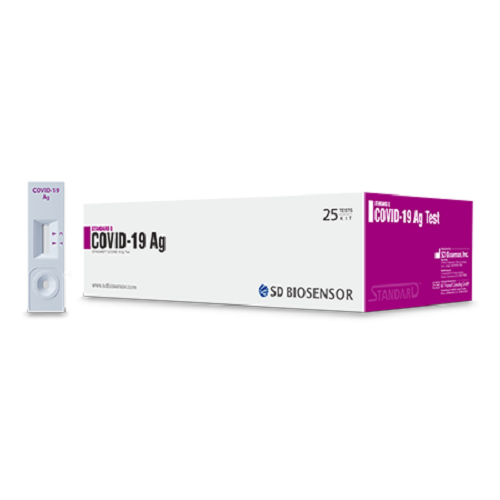
The SD Biosensor COVID-19 Rapid Antigen Test Kit (WHO, NCDC Approved) is a reliable, fast, and easy-to-use solution for detecting active COVID-19 infections. Sold by Neatstore, this high-quality test kit is ideal for individuals, clinics, workplaces, and public health organizations seeking Affordable infection detection products in Lagos without compromising accuracy.
For anyone looking to Buy SD Biosensor COVID-19 test kit Nigeria, this device provides convenience, speed, and professional-grade performance in a single package.
Quick & Accurate Results
Time is critical when controlling the spread of COVID-19. The SD Biosensor Rapid Antigen Test Kit delivers results within 15–20 minutes, allowing for timely detection and intervention. The kit uses nasal swab samples to identify the presence of SARS-CoV-2 antigens, helping limit exposure and reduce transmission risk.
Key benefits include:
-
✔ Fast and reliable results in minutes
-
✔ WHO and NCDC approved for safety and accuracy
-
✔ Easy-to-read cassette display
-
✔ Portable and convenient for home or professional use
-
✔ Suitable for repeated screenings
Complete & Easy-to-Use Kit
Each kit comes with all the necessary components for immediate testing, including:
-
Test cassette
-
Sterile nasal swab
-
Extraction buffer solution
-
Extraction tube
-
Step-by-step instruction manual
The intuitive design ensures both trained professionals and users at home can perform tests accurately with minimal effort.
Ideal for Multiple Settings
Whether for individual use, healthcare facilities, corporate workplaces, schools, or travel screening, this SD Biosensor kit is versatile and reliable. Its portability and simple operation make it perfect for:
-
Home self-testing
-
Hospitals and clinics
-
Workplace health programs
-
School and university screenings
-
Public health campaigns
Why Purchase from Neatstore
Neatstore guarantees authentic, high-quality products delivered directly to you. When you Buy SD Biosensor COVID-19 test kit Nigeria from Neatstore, you benefit from:
-
Certified WHO and NCDC-approved products
-
Competitive pricing for all budgets
-
Fast delivery within Lagos and nationwide
-
Bulk purchase options for organizations
-
Reliable customer support
Take Control of Health & Safety
Rapid testing remains a key element in managing infectious diseases. The SD Biosensor COVID-19 Rapid Antigen Test Kit empowers individuals and organizations to respond quickly, limit transmission, and maintain safe environments.
Order today from Neatstore and access one of the most trusted Affordable infection detection products in Lagos for effective COVID-19 screening.